
Laminar Airflow Ceilings for ISO 5 Orthopedic Surgeries:
Achieve Superior Sterility in Orthopedic Surgery with ISO 5 Laminar Airflow Ceilings: Protect Hip and Knee Implants from Contamination
The Operio Laminar Airflow System and SteriStay Instrument Table revolutionize sterility in orthopedic surgeries by ensuring ISO 5 ultra-clean conditions. Operio delivers targeted laminar airflow directly to the surgical site and instruments, protecting against airborne contamination and reducing the risk of surgical site infections (SSIs).
SteriStay complements this by extending the sterile zone with localized laminar airflow, safeguarding instruments and implants during preparation and surgery. Together, these portable, cost-effective solutions transform ISO 7 operating rooms into ISO 5 environments, ensuring superior sterility for procedures like hip and knee replacements without requiring structural modifications.
The Challenge: Limited Laminar Airflow Coverage in Orthopedic Surgery
In orthopedic operating rooms, maintaining sterility is critical, especially during procedures involving implants such as hip replacements and knee prostheses. However, many operating rooms are equipped with laminar airflow systems that are too small to provide comprehensive sterile coverage for both the operating field and the instrument preparation area.
Risk of Instrument Contamination
Instruments prepared outside the limited sterile zone of the laminar airflow ceiling are exposed to significant contamination risks. Airborne particles, often laden with bacteria, settle on instruments during preparation and surgery, increasing the likelihood of surgical site infections (SSIs). This is particularly problematic in orthopedic surgeries, where infections can lead to severe complications, prolonged recovery times, and costly treatments.
Gaps in Existing Laminar Airflow Systems
- Insufficient Coverage: Many airflow ceilings are too small to extend the sterile zone to both the surgical field and instrument area simultaneously.
- Airflow Disruption: Surgical lights, operating personnel, and equipment often create turbulence, further diminishing the protective effects of traditional airflow systems.
- ISO Standard Limitations: Operating rooms classified as ISO 7 lack the sterility required for high-risk orthopedic procedures, failing to meet ISO 5 standards critical for implant surgeries.
The Solution: Steristay Instrument Table
Steristay: Advanced Instrument Protection for Orthopedic Surgery
Steristay is an innovative instrument table equipped with localized laminar airflow, designed to ensure ISO 5 ultra-clean conditions for orthopedic surgeries. It extends the sterile zone, protecting instruments and implants from contamination during procedures like hip and knee replacements. Cost-effective and portable, Steristay upgrades ISO 7 operating rooms to ISO 5 standards without construction, significantly reducing infection risks.
Steristay offers a targeted solution:
- Extends Sterile Coverage: Expands the sterile zone beyond the existing airflow ceiling, providing continuous protection during preparation and surgery.
- Upgrades to ISO 5 Standards: Allows existing ISO 7 rooms to achieve ISO 5 conditions quickly, cost-effectively, and without construction.
Why Steristay?
1. Protects Instruments from Contamination
- Steristay creates a localized laminar airflow shield, eliminating 99.995% of bacteria and microorganisms using H14 HEPA filtration.
- Ensures sterility for instruments and implants from preparation to the end of surgery.
2. Cost-Effective and Ready to Use
- No Construction Needed: Operates independently of existing ventilation systems and requires no structural modificationsOrtopedia_2024.Lr.
- Affordable Solution: A fraction of the cost of traditional cleanroom upgrades.
3. Compatible with Existing Airflow Systems
- Integrates seamlessly into rooms with existing laminar airflow ceilings without creating turbulence
Scientific Evidence Supporting Steristay
- Reduces Contamination: Studies show a 95% reduction in bacterial contamination of instruments during orthopedic implant surgeries.
Operio Laminar Air Flow: Revolutionizing Sterility in Orthopedic Surgery
The Operio Laminar Air Flow System is a groundbreaking solution for orthopedic surgeries, providing unmatched sterility by delivering ultra-clean, ISO 5 air directly to the operating field. Whether for complex joint replacement surgeries or minor ambulatory procedures, Operio offers flexibility, cost-effectiveness, and improved operational efficiency.
Key Advantages of Operio Laminar Air Flow
1. Direct Sterile Air to the Operating Field
- Operio ensures ISO 5 ultra-clean air flows directly to the surgical site and instruments.
- Its innovative design bypasses common obstacles such as surgical lights and the heads of operating staff, maintaining uninterrupted sterile airflow.
2. Immediate, Plug-and-Play Solution
- Operio is a portable and ready-to-use device requiring no construction or major modifications.
- Simply plug it in to upgrade existing ISO 7 operating rooms to ISO 5 standards quickly and efficiently.
3. Ideal for Ambulatory Surgeries
- Operio enables ISO 5 air quality for minor ambulatory procedures such as:
- Hand surgery
- Foot surgery
- Intravitreal injections
- By shifting these procedures from operating rooms to ambulatory rooms, hospitals can maximize operating room availability for high-value surgeries.
4. Increased Operating Room Efficiency and Profitability
- Moving smaller procedures to ambulatory settings frees up operating rooms for critical surgeries like:
- Hip implant surgeries
- Knee prosthesis operations
- This optimization allows healthcare providers to perform more high-revenue procedures while reducing operational costs associated with extended operating room use.
5. ISO 7 to ISO 5 Upgrades: Revolutionize Your Operating Theatre with Advanced Sterility
Upgrading existing operating theatres from ISO 7 to ISO 5 cleanroom standards is now achievable with innovative solutions like the Operio Laminar Air Flow System. These upgrades provide ultra-clean environments essential for advanced surgical procedures, ensuring the highest levels of patient safety and infection control.
The Norwegian Case Study: Reducing Infection Rates with Operio and SteriStay
Revolutionizing Infection Prevention at Drammen Hospital, Norway
For the past three years, Operio and SteriStay mobile units have been instrumental at Drammen Hospital in Norway. These innovative devices complement the existing structural ventilation systems by significantly reducing the number of airborne bacteria, a known risk factor for open wounds and sterile instruments.
Key Results from the Norwegian Case Study
1. Infection Rates Before Operio and SteriStay
- Pre-implementation infection rate:
- The risk of infections was between 1.5–2%, a rate consistent with data from Scandinavian healthcare databases.
2. Infection Rates After Implementation
- Post-implementation results:
- Out of 5,000 surgeries for knee and hip replacements, the risk of deep infections dropped dramatically to 0.27%.
3. Conclusion
- The data highlights the critical importance of reducing intraoperative contamination to prevent deep infections, especially in procedures like knee and hip replacements, which are particularly prone to such complications.

Get in Touch with Normeditec
We’re here to help you enhance your surgical solutions with the Operio Mobile Laminar Air Flow Unit. Contact us for expert advice, product information, or assistance.
Contact Information
Email: info@normeditec.com
Phone:
- Germany: +49 7139 2090859
- Italy: +39 348 730 24 45
WhatsApp Support:
- Germany: +49 176 62 13 16 34
- Italy: +39 348 730 24 45
Languages Spoken: English, German, French, Italian
Website: www.normeditec.com
We deliver worldwide and are available via video call for support and guidance. Let’s make advanced aseptic solutions accessible to your practice today!
Benefits:
Prevention of periprosthetic infections
Instruments and implants can be prepared under aseptic cleanroom conditions
Enlarge of the protective zone in the operating theatre without turbulence
The Sterile Air Flow is experienced as very pleasant by personnel, because the air it produces is not cold and it is also very quiet – comparable to a projector
to upgrade existing operating and minor procedures rooms quickly and cost-effectively.
The units are extremely small and easy to handle
tracking systems for defensive medicine
Related products
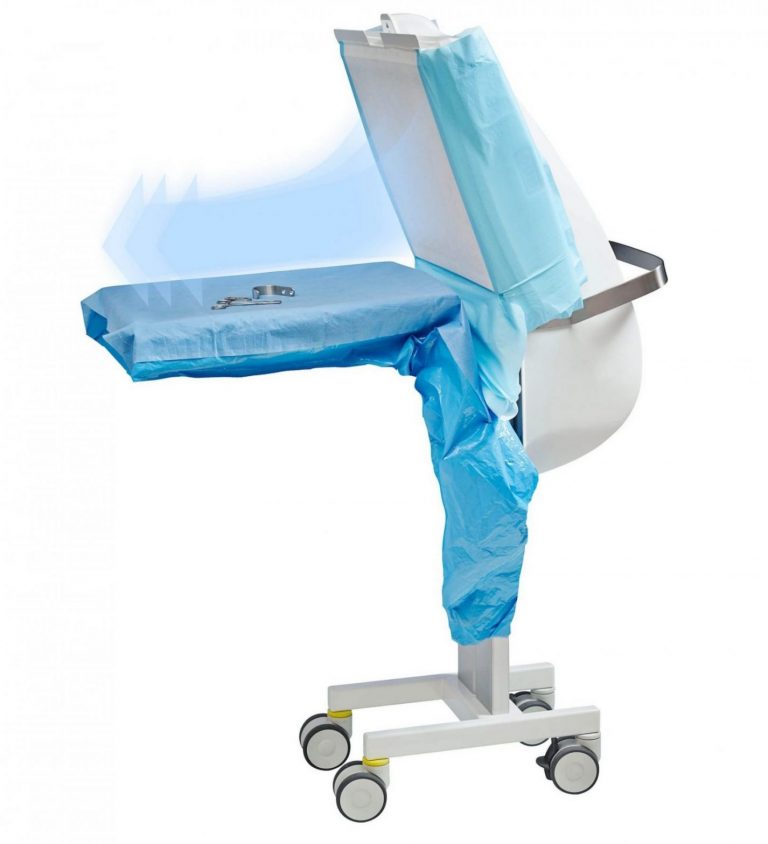
Operio portable laminar air flow
The Operio Mobile Laminar Air Flow Unit provides ISO 5 sterile conditions, transforming conventional operating rooms and outpatient surgical spaces into ultra-clean environments. Its focused sterile airflow protects the surgical field and instruments, ensuring maximum sterility for eye surgeries, ENT procedures, day surgeries, cardiology, all types of implant surgeries, plastic and aesthetic surgeries, orthopedic operations, and more. Ideal for enhancing infection control and surgical precision across diverse medical disciplines.
Go to product
Steristay instrument table with hepa filter
TOUL Steristay– Focused laminar flow with built-in backtable is ideal to keep the surgical instruments sterile. While implants are opened only at the last moment to ensure sterility, the instruments used for implantation remain exposed on traditional instrument tables. This exposure significantly increases the risk of contamination and surgical site infections.
Go to product
 it
it en
en fr
fr de
de












